#FEMSmicroBlog: World Tuberculosis Day and our fight against one of the most dreadful diseases
Tuberculosis has affected humans for thousands of years and remains the leading cause of death from a single infectious agent. According to the WHO, approximately one quarter of the global population is infected with Mycobacterium tuberculosis. In 2024, around 10.7 million new tuberculosis cases were reported globally, while approximately 1.23 million people died from this disease. However, in recent years, significant progress has been made in understanding how Mycobacterium tuberculosis exploits its host to survive and cause disease. New drugs, such as bedaquiline, delamanid, and pretomanid, have been introduced to shorten therapy and treat drug-resistant tuberculosis. For this year’s World Tuberculosis Day, Devendra Pratap Singh discusses a novel approach to fight this dreadful disease. #MicrobiologyEvents
Mycobacterium tuberculosis knows how to adapt thanks to its sensing machineries
Mycobacterium tuberculosis possesses remarkable adaptive capabilities, allowing it to persist within the host without causing active disease. During this phase, the bacterium enters a non-dividing, metabolically quiescent dormant state, known as latency and; this is responsible for latent tuberculosis infection.
Surviving inside host cells and establishing infection is possible due to the pathogen’s ability to sense and evade host defence mechanisms. One of the primary pathways employed by Mycobacterium tuberculosis for this involves a multitude of two-component signalling systems.
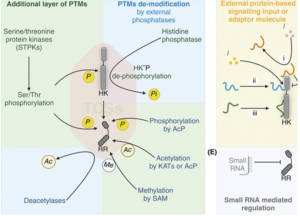
These pathways consist of two proteins: a histidine kinase, which senses environmental changes, and a response regulator, which generates adaptive responses. Activated by an environmental cue, the histidine kinase undergoes autophosphorylation and subsequently transfers a phosphate group to its cognate response regulator, which then modulates gene expression for an adaptive outcome.
Mycobacterium tuberculosis relies on multiple two-component signalling systems to regulate critical cellular processes and adapt to the dynamic host environment. These systems are essential for viability, bacterial growth, and replication, key metabolic regulations, including redox homeostasis, entry into dormancy, as well as persistent infection and reactivation leading to active disease.
Challenges and opportunities two-component signalling systems offer in tuberculosis research
Latent tuberculosis infection and drug-resistant tuberculosis remain major challenges, highlighting the need for new therapeutic targets. Recent efforts in anti-tuberculosis drug discovery focus on proteins that play crucial roles in bacterial adaptation within the host, particularly during latency, persistence, and reactivation.
Currently, two-component signalling systems are considered promising targets for novel anti-tuberculosis drugs. Pathways essential for Mycobacterium tuberculosis viability, dormancy regulation, adaptation and virulence hold significant promise as potential therapeutic targets. Examples include MtrAB and PrrAB for viability, DevRST for dormancy regulation, and PhoPR for adaptation and virulence. . By targeting and inhibiting these systems, it may be possible to compromise Mycobacterium tuberculosis’s ability to sense and respond to host-derived stresses, rendering it more vulnerable to host defences or treatments.
However, these efforts have not yet been successful. A primary reason for this is our incomplete understanding of these systems, specifically, what signal they sense, when they are activated, and how they orchestrate adaptive changes.
Another significant challenge is that these systems do not function in isolation as linear pathways. Instead, they emerged to function as highly interconnected networks.
This type of signalling design presents a major obstacle in targeting two-component signalling systems, as inhibiting one component function can be compensated by another. Therefore, effective targeting of these systems requires consideration of their interconnected design rather than a focus on individual components.
Looking ahead: sensing biology and the future of tuberculosis control
This novel concept is discussed in the review “Multiple input–multiple output (MIMO) designs in two-component signalling systems of Mycobacterium tuberculosis” published in FEMS Microbiology Reviews. The authors outline how a deeper understanding of the in vivo roles of Mycobacterium tuberculosis’s two-component signalling systems is still required for successful therapeutic interventions. Molecular insights into signalling mechanisms during infection could open new possibilities for interventions.
In all, integrating insights from host-related risk factors, improved therapeutics, diagnostics, and public health policies will be critical if we are to move closer to the goal of ending tuberculosis.
 Devendra Pratap Singh is currently a postdoctoral researcher at the Institut Pasteur, France. He completed his PhD from the Indian Institute of Science (IISc), Bangalore, India, where his research was focused on two-component signalling crosstalk and its physiological significance in Mycobacterium tuberculosis. Prior to this, he earned his master’s degree in Biochemistry from Dr. Rammanohar Lohia Avadh University, Ayodhya, India. During this period, he gained his first laboratory research experience at the CSIR–Indian Institute of Toxicology Research (IITR), Lucknow, India – an exposure that sparked his interest in research.
Devendra Pratap Singh is currently a postdoctoral researcher at the Institut Pasteur, France. He completed his PhD from the Indian Institute of Science (IISc), Bangalore, India, where his research was focused on two-component signalling crosstalk and its physiological significance in Mycobacterium tuberculosis. Prior to this, he earned his master’s degree in Biochemistry from Dr. Rammanohar Lohia Avadh University, Ayodhya, India. During this period, he gained his first laboratory research experience at the CSIR–Indian Institute of Toxicology Research (IITR), Lucknow, India – an exposure that sparked his interest in research.The section #MicrobiologyEvents for the #FEMSmicroBlog reports about events and meetings relevant to our network. These include world awareness days, FEMS-sponsored meetings or meetings of Member Societies and many more.
| DO YOU WANT TO BE A GUEST CONTRIBUTOR? |
| The #FEMSmicroBlog welcomes external bloggers, writers and SciComm enthusiasts. Get in touch if you want to share your idea for a blog entry with us! |



