The pathogen Brucella causes Brucellosis, a neglected zoonotic disease with a considerable impact on global health, especially in low- and middle-income countries. It spreads from animal hosts to humans and other mammals via close contact, where it creates a niche by manipulating host cell functions. The main tool for this process is the type 4 secretion system (T4SS), a nano syringe that injects effector proteins into the host cell. The study “A subset of type 4 secretion system effectors of Brucella spp. associates to outer membrane vesicles” in the Thematic Issue “Type IV secretion systems: structure, function, evolution, and role in conjugation or host pathogen interaction” in microLife identified a possible alternative secretion mechanism in the pathogen, as explained by Maren Ketterer in this #FEMSmicroBlog. #FascinatingMicrobes
The Brucella-type 4 secretion system-conundrum
To survive and replicate inside a host, Brucella specifically (but not exclusively) invades macrophages – the very cells that mammals employ to detect and destroy intruders. For this, Brucella allows itself to be ingested by macrophages but not digested and killed. Instead, it manipulates the cell to create a hospitable hide-out, converting the host’s cellular machinery into a bacterial nursery.
To create this niche, Brucella manipulates host cell functions using specialized tools, like the type 4 secretion system. Unlike other bacterial type 4 secretion systems, however, Brucella’s type 4 secretion system lacks a canonical coupling protein, essential for effector recognition and delivery to the machinery.
Furthermore, type 4 secretion system effectors are not essential to establish infection and do not have classical translocation signals that facilitate the interaction of the effector protein with the secretion machinery. In short, we don’t understand how this machinery recognizes its effector proteins. Additionally, the presence of type 4 secretion system-independent effector proteins indicates alternative secretion mechanisms.
Effector export via outer membrane vesicles instead of type 4 secretion
The study “A subset of type 4 secretion system effectors of Brucella spp. associates to outer membrane vesicles” in microLife identified outer membrane vesicles as an alternative export mechanism for some type 4 secretion system effectors.
For decades, the scientific community has regarded outer membrane vesicles as bacterial debris: tiny spheres made from the outer membrane, filled with proteins and other cargo. Yet these nanostructures are also important for host cell manipulation.
The study identified 13 type 4 secretion system effectors in purified outer membrane vesicles of Brucella abortus, a species causing human disease and abortions in cattle. As expected, outer membrane proteins such as Omp22 and BmaC were enriched, while inner membrane proteins such as BvrS and SecF were depleted in the outer membrane vesicle fraction.
The effector proteins NpeA, BspD, and BspC were also enriched, demonstrating specific packaging and sorting mechanisms and arguing against the “bacterial debris” theory. These outer membrane vesicle-associated proteins often possess functional signal peptides that direct them to the periplasm via the Sec-pathway before being packaged into outer membrane vesicles and secreted.
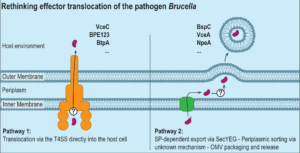
Furthermore, by using comparative proteomics across different Brucella species, the authors discovered that a subset of the type 4 secretion system effectors, namely VceA, BPE043, BspC, BspD, BspL, and NpeA, consistently localizes to outer membrane vesicles.
Overall, these results suggest an alternative secretion mechanism for some effectors that were previously claimed to be secreted by the type 4 secretion system: instead, or additionally, Brucella may utilize regulated vesicle biogenesis to export these proteins.
Future research should investigate if and how outer membrane vesicles are involved in the infection process and at which stage of the infection. Furthermore, it is important to understand which processes govern outer membrane vesicle biogenesis and cargo packaging. Answering these questions would significantly deepen our understanding of outer membrane vesicle biogenesis and Brucella-host interaction.
- Read the article “A subset of type 4 secretion system effectors of Brucella spp. associates to outer membrane vesicles” in the Thematic Issue “Type IV secretion systems: structure, function, evolution, and role in conjugation or host pathogen interaction” by Ketterer et al. in microLife (2026).

Maren Ketterer is a postdoctoral research fellow in the Group of Christoph Dehio at the Biozentrum, University of Basel (CH). She earned her MSc in Microbiology from the University of Bonn (GER) in 2017 and completed her PhD in Microbiology at the University of Basel in 2023. Her research during the PhD focused on Brucella-host interaction mediated through protein translocation. Maren’s long-term goal is to establish her own research group dedicated to uncovering the mechanisms that allow Brucella to persist and survive treatment within host cells, with the aim of informing more effective strategies to treat human Brucellosis.
About this blog section
The section #FascinatingMicrobes for the #FEMSmicroBlog explains the science behind a paper and highlights the significance and broader context of a recent finding. One of the main goals is to share the fascinating spectrum of microbes across all fields of microbiology.
| Do you want to be a guest contributor? |
| The #FEMSmicroBlog welcomes external bloggers, writers and SciComm enthusiasts. Get in touch if you want to share your idea for a blog entry with us! |



